As I’m sure most of you know, this has been a controversial topic for more than 2 decades. The problem is fairly simple: Screening can pick up earlier disease, save some lives, but treatment has side effects for virtually 100% of men who get treated, and “active surveillance” is not a picnic with repeat biopsies every 2-3 years. We may have to treat as many as ten men to save one life. On the other hand, if they live long enough, more than half of men probably develop prostate cancer, usually of the low grade (Gleason 6 or less) type that will never bother them. Here is a nice article that shows how autopsy series over time have found prostate cancer in up to half of men, dependent on age, race, etc. but notably pointing out how seldom autopsies are now performed compared to earlier eras. The reality is that we have no idea these days how many 90 year old men would have a small cancer if we really looked hard for it. What we understand is that they didn’t need to know they had a prostate cancer if they were never treated and died from something else.
Now, add to these challenges the revolution in cancer detection provided by molecular testing. This field is moving so fast that the “old idea” of PSA screening is becoming passé. For example, Illumina, the company that makes automated next generation sequencing machines spun off a startup, GRAIL that developed a “pan-cancer” test that looks for fragments of DNA circulating in the blood, the fingerprints for most of the common cancers. This test, called “Galleri” is undergoing real world testing in the UK, but is not covered or approved in the U.S. Proponents (some of whom are consultants for biotech companies) suggest that it could save “millions of lives”. The test, because we live in a free, capitalistic society is already marketed on the internet for an out-of-pocket price of only $949 with payment plans available. But…and the prostate cancer community knows this perhaps better than any other…the challenge of knowing whom to test, when to test, and what to do with a positive test may take decades to figure out. Here’s an article covering some of those promises and challenges (false positives, lead time bias, costs for treatment, etc.)
But for prostate cancer, the same DNA technology is making real progress. What we want are tests that not only tell us who has prostate cancer, but who has the kind of cancer that NEEDS to be treated or followed closely, and lowers the detection of clinically insignificant cancers. An example of this kind of testing sophistication appeared in NEJM this month from a group in Stockholm. This group has developed a test called Stockholm3 that is “a risk-prediction model that is based on clinical variables (age, first-degree family history of prostate cancer, and previous biopsy), blood biomarkers (total PSA, free PSA, ratio of free PSA to total PSA, human kallikrein 2, macrophage inhibitory cytokine-1, and MSMB), and a polygenic risk score (a genetic score based on 254 single-nucleotide polymorphisms [SNPs] and an explicit variable for the HOXB13 SNP) for predicting the risk of prostate cancer with a Gleason score of 7 or higher.” They then took men at risk of having prostate cancer (PSA>3 and Stockholm3 >11%) and either did “blind” 12 core biopsies or did an MRI first and included targeted biopsies of high risk lesions only if seen on the MRI.
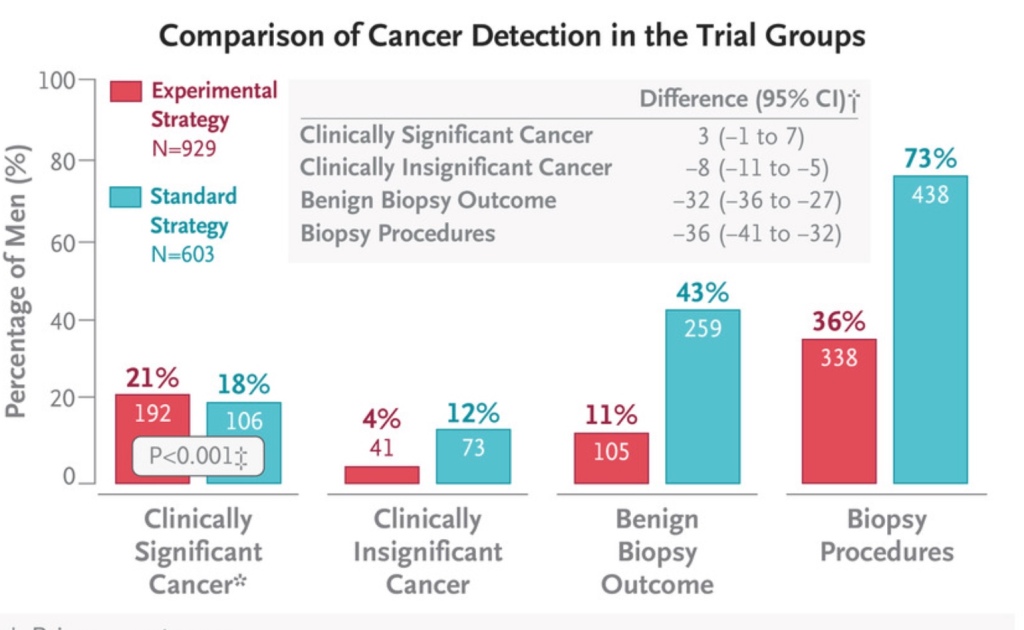
Note that the number of biopsies needed went down, as did the number of benign or clinically insignificant cancers. This is the sort of effort that will eventually reduce the number of men having unnecessary biopsies or treatment by combining all of the great new molecular and radiology technologies (dynamic contrast enhanced MRI’s). We now routinely use some of the molecular tests to help us in screening and deciding about treatments as I reviewed in this blog.
While we are still a long way from applying this kind of technology to “every man over 50”, the future for the next generation (our sons and grandsons) will be much better – fewer unnecessary biopsies and treatments. Hopefully this type of approach can be applied to the pan-cancer type of “Galleri” screening being proposed, and make such testing cost effective as well. Congratulations to the prostate cancer researchers and their patients for leading the way!




Excellent. I just received news that my PSA is slightly above the norm and have thus scheduled an appointment with a urologist. I now better understand how to talk to him.
BD